 Refer to our Chemical Reaction article for additional facts and information providing the different types of reactions, examples of reaction and the Rate of a Chemical Reaction. There are several different types of Chemical reaction which have been detailed below: Some examples of a chemical reaction include most commonly burning, fermentation, tarnishing and rusting. Aluminium reacts with dilute nitric acid to form aluminium nitrate and hydrogen gas.Aluminium reacts with dilute sulphuric acid to form aluminium sulphate and hydrogen gas.At high temperatures, aluminum reacts with nitrogen, carbon, and sulfur, forming aluminum nitride, carbide and sulfide.Iodine reacts with aluminum during heating.Chlorine and liquid bromine react with aluminum at room temperature.Aluminium reacts with dilute hydrochloric acid to form aluminium chloride and hydrogen gas.Aluminum chloride reacts with lithium hydride to form Lithium aluminum hydride.Aluminum reacts with Hydrogen to form Aluminum hydride.The ability of aluminum to combine actively with oxygen is used in the reduction of metals from their oxides.Įxamples of Aluminum Reactions are as follows: Strong heating is required to make aluminium powder burn in oxygen and a white powder is formed. During combustion of aluminum in an oxygen current, a temperature of over 3000☌ is reached. 
Powdered Aluminum powdered burns energetically in air. The product is aluminum chloride, a white powder that fumes in air, reacting with water to form HCl gas and aluminum oxide. It fumes in moist air and hisses when mixed with liquid water. Very toxic chemicals, or those that are reproductive hazards or sensitizers, require expert advice on control measures if a less toxic chemical cannot be. Aluminum and chlorine do not react rapidly at room temperature, but if the aluminum is heated, the reaction is vigorous. Pure samples are white, but sometimes it has a yellow colour. This chloride is used as a wood preservative or catalyst.Īluminium + Steam -> Aluminium oxide + HydrogenĪluminum Chloride is a compound of aluminium and chlorine that has a very high affinity for water. A strong layer of aluminium oxide coats the metal which sometimes prevents the reaction with steam to give aluminium oxide and hydrogen gas. 
Most of the aluminum salts dissolve well in water. It is also used in the dehydration of alcohols to alkenes.The properties of Aluminum include low density, high thermal conductivity, high electrical conductivity, high plasticity and high resistance to corrosion.Īluminum reacts explosively with water but does not react with cold water. 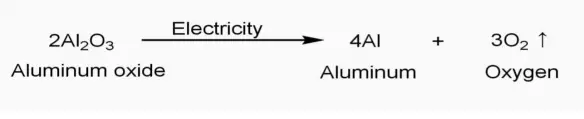
It is used as a catalyst in the Claus process which is the conversion of hydrogen sulfide waste gases into elemental sulfur. For example, aluminosilicate glass contains 5 to 10 percent alumina or aluminium oxide.Īluminium oxide also acts as an industrial catalyst. Many different kinds of glass have aluminium oxide as a common constituent. 
It is a common ingredient in sunscreen and is also present sometimes in cosmetics like nail polish, lipstick and blush. Hint: Aluminium oxide or aluminum oxide is the name of a chemical compound containing aluminium and oxygen.Īluminium oxide has the chemical formula $$ which is then converted into aluminium by the electrolysis of mixture of alumina and cryolite via the Hall and Heroult process.Īluminium oxide is used as a filler for plastics as it is chemically inert and white. 
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
